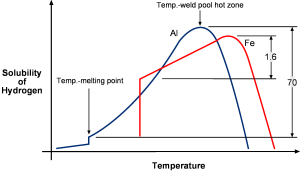
Porosity in aluminum welds is caused by bubbles of hydrogen gas which are trapped in the welds as it cools. The source of this hydrogen is oils, greases, or water vapor that is dissociated by the welding arc. In order to control weld porosity, the sources of these contaminates must be eliminated.
In its solid state, aluminum is extremely resistant to hydrogen as a result of the oxide layer. However, in its molten state, aluminum is extremely hydrogen soluble, making it more susceptible to porosity than steel.
Porosity can usually be eliminated by taking the following precautions:
- Remove oils and greases from the surface of the base material.
- Wiping with a clean rag saturated with a degreasing solvent. This method is very effective.
- Mild alkaline solutions make good degreasers. The part to be degreased can be sprayed with these solutions or dipped into a tank containing them. Since such cleaners are usually water based, it is important to thoroughly dry the part after degreasing.*
- Acid based cleaning solutions for cleaning aluminum. These are usually effective. However, all are acidic and some contain hydrofluoric acid, so caution in their use and disposal is required. Again, since they contain water, the piece must be thoroughly dried before welding.*
- Remove excess oxides.
- Brush lightly with a stainless steel wire brush.
- Oxide removal can also be performed by immersing the part in a strong alkaline solution.
- Interpass cleaning with degreaser and/or wire brush.
- Remove oxides and weld smut (composed largely of aluminum oxide and magnesium oxide) before depositing another pass with a stainless steel wire brush using light pressure.
- When welding in the vertical position, use an upward progression, rather than the downward progression usually used to weld steel, to minimize weld porosity.
Tips for Troubleshooting porosity
- Make sure the base material is free of moisture.
- Be wary of temperature variations i.e. bringing cold plate into a warm shop can cause condensation on the base material.
- Protect the spool of wire. Use wire reel covers when possible. Store unused wire in climate controlled conditions in the manufacturer’s original packaging. Re-sealable bags are a plus!
- Be on the lookout for possible condensation in bulk shielding gas lines.
- Provide regular maintenance on water-cooler systems.
- Check for sources of contamination from the tools you use i.e. damp welding gloves, moisture in air lines used for air tools, dirty wire brush or wire wheel.
- Grinding discs always use organic binding compounds. Be wary of changing disc manufacturer as you may introduce hydrocarbon’s from a grinding disk. Look for aluminum specific discs.
Copper contamination will also appear white on a radiograph. Copper contamination is usually larger and “fuzzier” than tungsten, which appears as individual small particles.
Copper contamination is often encountered in GMAW and is caused when the wire burns back and fuses to the copper contact tip. The copper and aluminum quickly alloy and deposit copper in the aluminum weld. If this occurs, the copper contamination must be ground out and repaired because the aluminum/copper alloy that is deposited is very brittle.
Copper contamination can also be caused by copper backing bars or copper tooling. While the use of copper backing bars is acceptable, the joint preparation must be such that the arc is not allowed to impinge directly on the copper bar. If it does, the copper bar will melt and alloy with the aluminum. While copper backing bars are acceptable, wide root openings must be avoided.
Porosity verified with break test.
Porosity-free weld verified with break test.
*Always check local municipal by-laws and regulations before using a degreaser or acid cleanser.